From Depletion to Gradual Repair, Ozone Layer on Track for Recovery!
Over the decades, increasing global temperature has damaged the ozone layer that protects the earth from the ultraviolet radiation of the sun. Ultraviolet radiation damages living cells by mutating their DNA, and can also cause the proteins that give cells their structure to unravel and misbehave. To add to this solar radiation can also cause sunburns and skin cancer in humans.

A graphical representation of a hole in the ozone layer over the Arctic | Image: ESA
Ozone is a gas made up of a trio of oxygen atoms. In the stratosphere, about 12 to 18 miles about Earth’s surface, a thin layer of ozone absorbs a portion of the ultraviolet radiation emanating from the sun.
Over the years, human activities have resulted in the emission of many harmful gases, increased global temperature and changing climatic patterns. These changes have depleted this protecting layer.
Depletion of Ozone Layer
In 1974, Mario Molina and Sherwood Rowland, two chemists at the University of California, Irvine, published an article in Nature detailing threats to the ozone layer from chlorofluorocarbon (CFC) gases. At the time, CFCs were commonly used in aerosol sprays and as coolants in many refrigerators. As they reach the stratosphere, the sun’s UV rays break CFCs down into substances that include chlorine.
This research revealed that the thin layer had a finite capacity for absorbing chlorine atoms in the stratosphere. As per the US Environmental Protection Agency, one atom of chlorine can destroy more than 100,000 ozone molecules, exterminating ozone much faster than it can be replaced.
In 1985, a team of English scientists found a hole in the ozone layer over Antarctica that was linked to CFCs. The “hole” is actually an area of the stratosphere with extremely low concentrations of ozone that reoccurs every year at the beginning of the Southern Hemisphere spring (August to October).
The ozone hole first appeared over Antarctica because atmospheric and chemical conditions unique to
this region increase the effectiveness of ozone destruction by reactive halogen gases. In addition to an abundance of these reactive gases, the formation of the Antarctic ozone hole requires temperatures low enough to form polar stratospheric clouds (PSCs), isolation from the air in other stratospheric regions, and sunlight.
While the global communities worked hard to repair this damage, another hole has made its presence felt over the Arctic. Scientists from the German Aerospace Center have recently spotted an unusually strong drop in ozone levels over the Arctic. Since March 14, levels have plummeted to less than 220 Dobson Units, which constitutes an “ozone hole.”
The hole has likely set a new scale for the largest ozone hole ever recorded in the North Pole. However, even though it is three times the size of Greenland, the tear is not likely to last long or be a danger to human health.
According to NASA researchers, ozone levels above the Arctic reached a record low for March. An analysis of satellite observations reveals that ozone levels reached their lowest point on March 12 at 205 Dobson units.
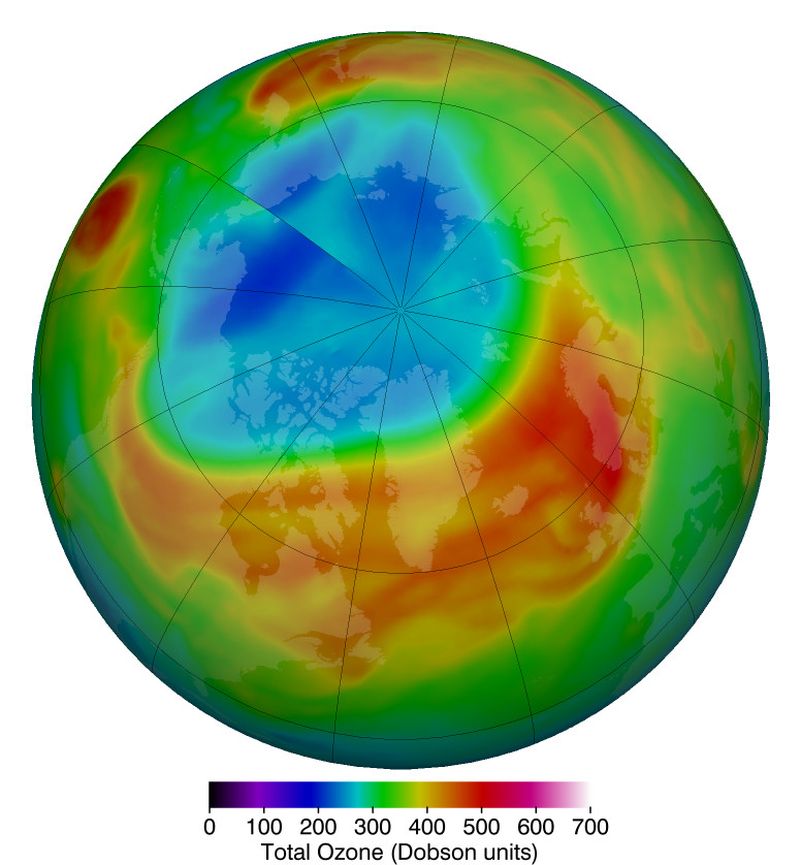
Arctic stratospheric ozone reached its record low level of 205 Dobson units, shown in blue and turquoise, on March 12, 2020 | Image: NASA
While such low levels are rare, they are not impossible to witness. Similar low ozone levels occurred in the upper atmosphere, or stratosphere, in 1997 and 2011. Comparatively, the lowest March ozone value observed in the Arctic is usually around 240 Dobson units.
The March Arctic ozone degradation was caused by a combination of factors that arose owing to unusually weak upper atmospheric “wave” events from December through March. These waves lead movements of air through the upper atmosphere similar to weather systems that are experienced in the lower atmosphere, but much bigger in scale.
New research published in the journal Geographical Research Letters analyzed Arctic ice and found an unintended consequence of the Montreal Protocol. The compounds that replaced CFCs have been released in the atmosphere, depositing far from their sources. The new generation of chemicals that replaced Freon, but are used in refrigerators, air conditioners and new cars have been accumulating since 1990.
A team of scientists at the University of Southampton has revealed that an extinction event 360 million years ago, which killed many flora and freshwater aquatic species on the earth, was caused by a brief breakdown of the ozone layer. This newly discovered extinction mechanism has deep implications for the current global warming trends.
With evidence showing that it was high levels of UV radiation, which collapsed forest ecosystems and wiped off many species of fish and tetrapods at the end of the Devonian geological period, 359 million years ago. This catastrophic burst of UV radiation happened as part of one of the earth’s climate cycles, rather than being caused by a huge volcanic eruption.
This event majorly happened owing to the rapidly warmed up climate following an intense ice age. Now, the researchers fear that the earth could be moving toward such an impending doom, with high temperatures that could possibly trigger a similar event.
Efforts to Repair the Damage
Recognition of the harmful effects of CFCs and other ozone-depleting substances led to the Montreal Protocol on Substances That Deplete the Ozone Layer in 1987. It was a landmark agreement to phase out substances signed by all 197 UN member countries.
Without the pact, the US would have seen an additional 280 million cases of skin cancer, 1.5 million skin cancer deaths, and 45 million characters- and the would be at least 25 percent hotter.
Almost 30 years later, NASA scientists documented the first direct proof that Antarctic ozone is slowly recovering as a result of the Montreal Protocol. Ozone depletion in the region has declined around 20 percent since 2005.
Also Read: Healing Ozone Layer is Redirecting Wind Flow Across the Globe
As CFC concentrations in the skies have deteriorated, the hole in the Southern Hemisphere’s ozone has contracted. But the ozone layer’s thickness over Antarctica remains certainly seasonal, and every year the region’s extremely cold winters cause the ozone layer above to wane. The dropping temperatures give rise to high-altitude clouds suffused with the CFCs still spinning in Earth’s atmosphere, which then rasp away at the nearby ozone.
At the end of 2018, the UN confirmed in a scientific assessment that the ozone layer is recovering, projecting that it would heal completely in the (non-polar) Northern Hemisphere by the 2030s, followed by the Southern Hemisphere in the 2050s and polar regions by 2060s.
What needs to be done?
While the recovery may not be as straightforward as expected, monitoring of the ozone layer is still going on. A study in early 2018 revealed that ozone in the lower stratosphere unexpectedly and curiously has dropped since 1998, while another study noted possible ongoing violations of the Montreal pact.
Whereas the world has decided that the use of hydrochlorofluorocarbons (HCFCs) and its transitional substitutes are harmful to the ozone layer, mankind is yet to part ways from these substances.
Developing countries need funding from the Montreal Protocol’s Multilateral Fund to eradicate the most widely used of these products, the refrigerant R-22. The next generation of coolants, HFCs, do not deplete ozone, but they are powerful greenhouse gases that trap heat, contributing to climate change.
Although HFCs represent a small fraction of emissions compared with carbon dioxide and other greenhouse gases, their planet-warming effect prompted an addition to the Montreal Protocol, the Kigali Amendment, in 2016. The amendment, which came into force in January 2019, aims to slash the use of HFCs by more than 80 percent over the next three decades.
Meanwhile, companies and scientists are working on climate-friendly alternatives, including new coolants and technologies that reduce or eliminate dependence on chemicals.


